CONTACT PRODUCT SPECIALIST
TECHNOLOGY IN DETAIL
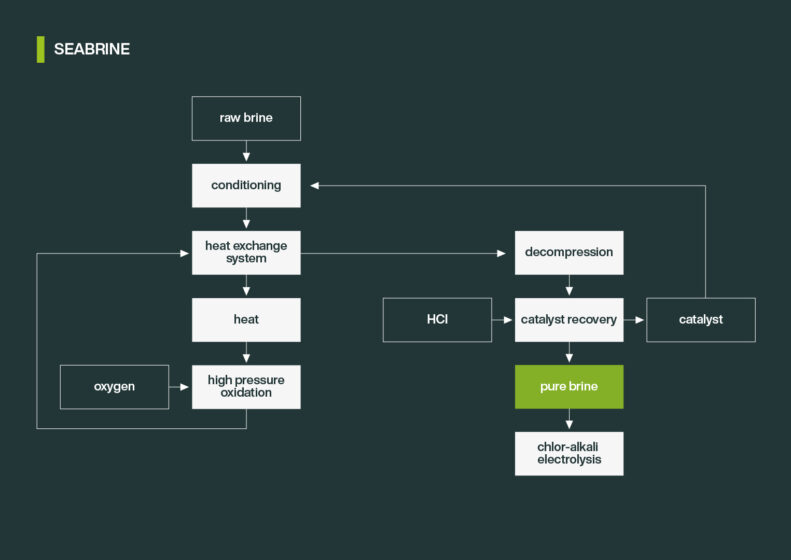
download SEABRINE flow diagram
The brine treatment process was originally developed by our engineering team to purify waste brine from epichlorohydrin and epoxy resin production, for reuse as raw material in other processes such as C/A electrolysis.
Contaminated waste brines typically consist of sodium chloride up to saturation concentration and partially chlorinated organic compounds (hydrocarbons, oxygen, nitrogen, and chlorinated compounds) which make it toxic to biological wastewater treatment plants.
Our advantage of treating wastewater to purity levels demanded by C/A electrolysis operators is unique.
The SEABRINE technology provides maximum energy recovery and low to zero waste. The process is divided in the following major steps:
Brine Pre-treatment
The pH-value is adjusted with caustic soda or hydrochloric acid. Typical pre-treatment steps are filtration and AOX reduction to prevent plugging and corrosion, respectively. Sodium chloride brines are additionally purified from heavy metal to meet the limiting values for electrolysis. Further pre-treatment steps are installed according to the raw brine contamination and client requirements.
High Pressure Oxidation
The oxidation is catalysed by a homogenous catalyst. The catalyst is mixed with the brine and the oxidizing agent. The brine is pressurized and preheated in a recuperator recovering the heat of the purified brine downstream of the reactor. The final operating temperature is realized by the exothermic oxidation reaction. During the oxidation organic compounds react to CO and CO2, hydrogen chloride, and ammonia. The purified brine is depressurized, and the heat is recovered for preheating. Depending on the organic compounds in the raw brine, a further treatment of the gas phase or the brine is implemented.
The oxidation rate is >99 %. The brine after the oxidation unit has a TOC value of <7 mg/l, which is within the specification for C/A electrolysis, particularly the strict limitations of membranes used.
Catalyst Recycling Unit
The homogenous catalyst is separated from the brine to close the catalyst cycle. The pH-value is adjusted, and the catalyst is removed from brine via sedimentation or filtration and ion exchange up to a limit of < 50 ppb, brine is treated via sedimentation and ion exchange up to a limit of <0,1 mg/l. The catalyst from both steps is treated and recycled back to the raw brine for oxidation.
The SEABRINE technology has a closed cycle regarding energy and chemicals. The excess heat of oxidation reaction is used to preheat the raw brine to reach the start temperature for the oxidation reaction. The catalyst runs through a closed loop cycle. Sodium hydroxide and hydrogen chloride can be produced from sodium chloride in the C/A electrolysis.
Key Advantages
- Save costs for crude salt and its logistics
- Reduce costs for crude brine cleaning
- Reduce costs for waste water treatment
- Low TOC values
- Reuse of purified sodium chloride brine in C/A electrolysis
- Closed loop catalyst cycle
- Recovery of hydrogen chloride and sodium hydroxide when combined with C/A electrolysis
Our customers include:
- Epichlorohydrin producers
- Epoxy-Resin producers
- Propylenoxide producers
- Glycerine producers
- Electrolysis plants, operating with membrane technology and using purified brine coming from other industrial process as raw material feed
- Other industries producing contaminated brine that need to be purified
References
Renewables
Innovative technologies for the production of sustainable chemicals from renewable feedstocks on eco-friendly closed loop concept
learn moreWet Sulfuric Acid
Production, recovery and regeneration of sulfuric acid from off-gases, spent acid or sulfur burning.
learn more
